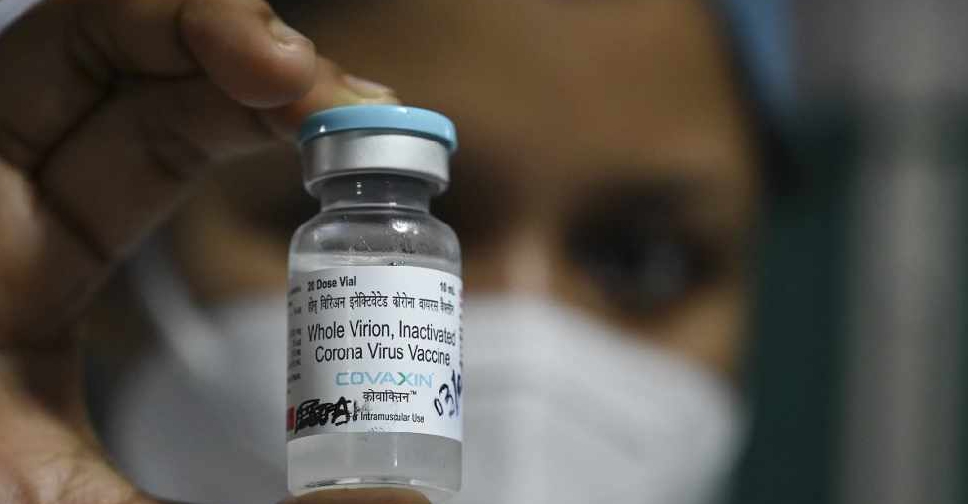
Bharat Biotech's homegrown COVID-19 vaccine has shown an interim vaccine efficacy of 81% in late-stage clinical trials, the Indian company said on Wednesday.
The interim analysis was based on 43 recorded cases of COVID-19 in the trial of 25,800 participants, conducted in partnership with the Indian government's medical research body.
Thirty-six of the 43 cases were recorded in participants who received a placebo, compared with seven cases in people who were given the Bharat Biotech vaccine, pointing to an efficacy rate of 80.6%, the company said.
India had approved the vaccine, branded COVAXIN, in January without late-stage efficacy data, raising questions about its effectiveness.
India's vaccination drive, currently underway, includes COVAXIN and a vaccine developed by Oxford University and AstraZeneca.
Earlier this week, Indian Prime Minister Narendra Modi was inoculated with the first dose of COVAXIN.



 Israeli military changes initial account of Gaza aid worker killings
Israeli military changes initial account of Gaza aid worker killings
 Two British lawmakers detained by Israel are travelling home
Two British lawmakers detained by Israel are travelling home
 Thousands in US and Europe protest against Trump, Musk
Thousands in US and Europe protest against Trump, Musk
 Israeli attacks on Gaza killed 60 people in 24 hours
Israeli attacks on Gaza killed 60 people in 24 hours



